Bhagtawala, Amritsar, Punjab
- GST NO. : 03AAGCT2565E1ZL
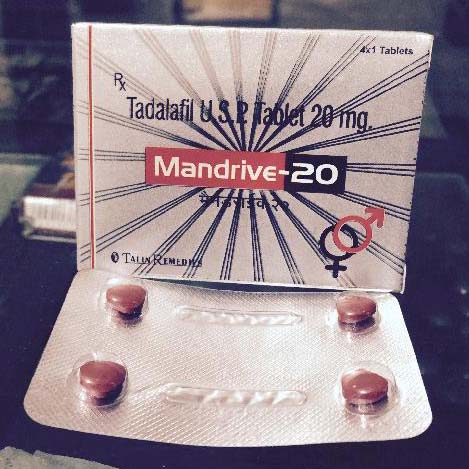
Mandrive Tablets
| Business Type | Manufacturer, Exporter, Supplier, Retailer, Wholesaler |
| Brand Name | Mandrive-20 |
| Generic Name | Tadalafil 20 mg |
Preferred Buyer From
| Location | Worldwide |
Mandrive for ED+BPH (erectile dysfunction + symptoms of benign prostatic hyperplasia)
Pharmacology : Inhibition of phosphodiesterase type 5 (PDE5) by tadalafil increases the concentrations of cyclic guanosine monophosphate (cGMP), resulting in relaxation of pulmonary vascular smooth muscle cells and vasodilation of the pulmonary vascular bed.
Mandrive-20 : Enhances the effect of nitric oxide at the nerve ending and endothelial cells in the corpus cavernosum by inhibiting PDE5 in the corpus cavernosum of the penis. This results in vasodilation, increased inflow of blood into the corporus cavernosum, and ensuing penile erection upon sexual stimulation.FDA Drug Approvals 2013
Pharmacokinetics
- Absorption
- Bioavailability undetermined. T max is 2 to 8 h.
- Steady state is attained within 5 days of once-daily dosing.
Distribution : Tadalafil is 94% protein bound
Metabolism : Predominantly metabolized by CYP3A4 to a catechol metabolite, which undergoes further metabolism to the major circulating metabolite, methylcatechol glucuronide.
Elimination : The terminal half-life is 15 h to 17.5 h. Tadalafil is excreted predominantly as metabolites, mainly in the feces (approximately 61%) and to a lesser degree in the urine (approximately 36%).
Special Populations :
- Renal Function Impairment : Tadalafil exposure (AUC) doubled in subjects with CrCl 31 to 80 mL/min. In patients with ESRD on hemodialysis, there was a 2-fold increase in C max and 2.7- to 4.8-fold increase in AUC.
- Hepatic Function Impairment : Tadalafil exposure (AUC) after a 10 mg dose in patients with mild or moderate hepatic impairment (Child-Pugh class A or B) was comparable with exposure in healthy subjects. There are insufficient data for subjects with severe hepatic impairment.
- Elderly : Subjects older than 65 y have a 25% higher exposure compared with subjects 19 to 45 y of age.
- Children : Tadalafil has not been evaluated in patients younger than 18 y.
- Gender : No clinically relevant differences in AUC and C max have been observed.
- Race : No differences in the typical exposure to tadalafil have been identified among different ethnic groups.
- Diabetes mellitus : AUC was reduced approximately 19% and C max was 5% lower in patients with diabetes mellitus than in healthy subjects.
- Indications and Usage : Treatment of pulmonary arterial hypertension ; treatment of erectile dysfunction, treatment of benign prostatic hyperplasia , and the treatment of combined erectile dysfunction and BPH.
Unlabeled Uses : Raynaud phenomenon.
Contraindications : Administration with organic nitrates (regularly and/or intermittently); serious hypersensitivity to tadalafil.
Dosage and Administration :
- Benign Prostatic Hyperplasia (With or Without Concomitant Erectile Dysfunction)
- Adults
- PO 5 mg once daily, taken at approximately the same time every day without regard to sexual activity.
Pulmonary Arterial Hypertension :
- Adults
- PO 40 mg once daily.
Erectile Dysfunction :
- Adults For use as needed
- PO 10 mg prior to anticipated sexual activity. The dose may be adjusted to 5 or 20 mg based on efficacy and tolerability. The max recommended frequency is once per day for most patients.
Once-daily use : PO Start with 2.5 mg once daily, taken at approximately the same time each day, without regard to timing of sexual activity. The dosage may be increased to 5 mg daily based on individual efficacy and tolerability.
Looking for "Mandrive Tablets" ?
Explore More Products